Show Me the Money! Finding Funding to Study the Clinical Effects of Mind and Body Interventions
April 19, 2017
At NCCIH, we are working hard to let the research community know about our new approach to funding research with human subjects that focuses on the clinical effects of complementary health approaches. We held a webinar yesterday to discuss why our Center will no longer accept most clinical trial applications through the Parent R01 and will, instead, encourage applicants to use a new series of funding opportunity announcements (FOAs) designed for investigator-initiated clinical trials (see NOT-AT-17-006). In this blog post, I’d like to share how our new FOAs provide pathways to support developmental steps for mind and body clinical trials.
Each of the clinical trial FOAs is targeted to a specific stage of clinical research. We want to help investigators select the appropriate FOA based on the amount of preliminary data that is available on the intervention’s effect in the patient population of interest. There are FOAs for early phase trials, intermediate trials, and full-scale efficacy, effectiveness, or pragmatic trials. The new series of FOAs will support investigators working to establish “building blocks” that bridge the gap from basic research to high-impact clinical trials on complementary and integrative health interventions.
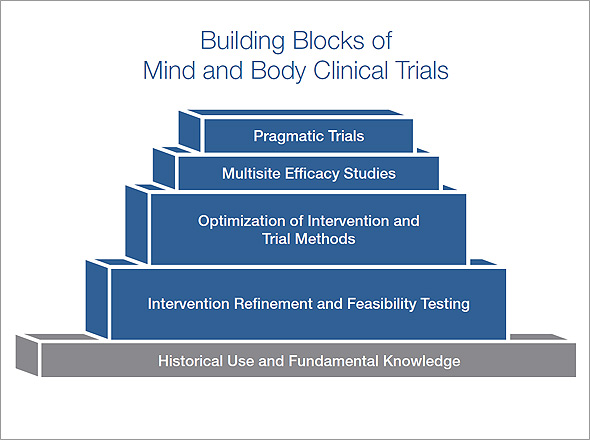
This graphic illustrates some of the building blocks that need to be addressed before conducting multisite efficacy or effectiveness studies.
- As shown at the base of the graphic, investigators should consider historical use of the intervention in humans and observational studies of the intervention’s effect. There may be a long history of use of the intervention, as in the case of yoga or acupuncture. Fundamental scientific knowledge about an intervention may exist, such as the impact of massage on fascia.
- The next level of the graphic depicts primarily early phase research that can be supported through the R34 FOA (PAR-14-182). This exploratory research can focus on refining the intervention to enhance feasibility and adherence, selecting the most appropriate outcomes to measure the impact of the intervention, and identifying the patient population that is appropriate for the future clinical trial.
- The third level from the bottom includes building blocks to determine whether the intervention can modulate a biological mechanism/psychological process in humans, and can be supported by the first phase of the R61/R33 FOA (PAR-17-149).
- The third level from the bottom also includes the need to optimize the impact of the intervention on the biological mechanism or psychological process by modifying the intervention, combining it with another treatment, or applying the intervention in a mechanism-relevant population. This “optimization” work can be supported by the second phase of the R61/R33 or the R33 FOAs (PAR-17-149 or PAR-17-162).
- Finally, the third level includes the need to establish the feasibility of a future clinical trial by demonstrating efficient methods for recruitment and retention of participants; complete data collection; fidelity of the intervention delivery across multiple sites; and appropriate data management methods. These additional elements can be gathered in an application to the U01 FOA (PAR-17-215).
Once a solid foundation of building blocks exists, researchers will be prepared to conduct multisite efficacy, effectiveness, or pragmatic trials. These multisite trials can be supported by the paired UG3/UH3 and U24 FOAs (PAR-17-175 and PAR-17-173).
Learn more about the building blocks of mind and body clinical trials and how to determine which FOA is right for your mind and body intervention study during our next webinar:
New NCCIH Funding Opportunities for Mind and Body Clinical Trials
Monday, April 24, 2017 (2:00 – 3:30 pm ET)

Comments
Comments are now closed for this post.